Disc Replacement Options Explained
THE BACKBONE
OF YOUR BUSINESS
ARE YOUR BOOKS
This "Quality of Motion" is a major patient benefit not available in any other implant!
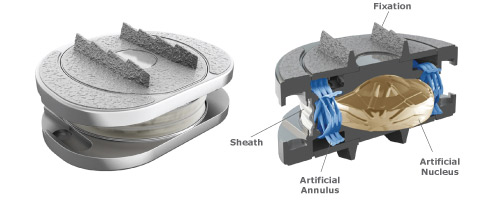
The Spinal Kinetics M6 Lumbar artificial disc replacement offers an innovative option compared to other artificial lumbar disc replacement because of its unique design, which is based on the qualities of the natural disc.
The M6 Lumbar Disc Replacement is the only artificial disc that incorporates an artificial nucleus (made from polycarbonate urethane) and a woven fiber annulus (made from polyethylene). The M6-L artificial nucleus and annulus are designed to provide the same physiologic motion characteristics of a natural disc. Extensive biomechanical testing with the M6-L artificial lumbar disc has demonstrated equivalent Quality of Motion compared to the healthy disc.
Together, the M6 Lumbar’s artificial nucleus and annulus provide compressive capabilities and a controlled range of natural motion in all 6 degrees of freedom. This “natural” motion is designed to provide the freedom to move your back naturally.
The M6 Lumbar Disc Replacement has two titanium outer plates with keels for anchoring the disc into the bone of the vertebral body. These outer plates are coated with a titanium plasma spray that promotes bone growth into the metal plates, providing long-term fixation and stability of the disc in the bone.
Many people ask what are the disc replacement options available?
Disc replacement was invented in the 80s in Germany at the Charite Hospital in Berlin.
The first disc replacement implant was the Charite Lumbar Disc Replacement.
Other hard plastic core and all metal disc replacement implants were developed over the years.
These early implants had some drawbacks and serious concerns!
- No shock absorption - creating impact damage risks
- Hyper Activity - creating wear on the facet joints
- Implant Migration - The implant moving into the vertebrae or forward or backwards
- Instability - Complicating multi-level use
Many of these implants are still in use despite these known risks.
Disc Replacement surgery options in the USA are limited by the FDA and Insurance carriers.
This means that patients who may be candidates for disc replacement from a medical point of view are often not offered a disc replacement surgery option in the US.
These patients are offered fusion surgery or are treated with less effective micro discectomy and laminectomy, sometimes called laser spine surgery, or older implant designs.
Newer advanced implant options resolved these issues and now provide shock absorption, better stability, motion control and improved end plate fixation.
The most advanced of these new disc replacement devices is the Spinal Kinetics M6 Disc Replacement.
Now available for the lumbar and cervical spine and for multi-level disc replacement surgeries



